

Stem Cell taking out the trash. Original Illustration from Emma Vidal of DrawImpacts. Credit: Emma Vidal
TOPICS: AgingCell BiologyMolecular BiologyStem Cells UCSD
By UNIVERSITY OF CALIFORNIA – SAN DIEGO MAY 11, 2023
Scientists at UC San Diego have discovered that stem cells’ unique method of discarding misfolded proteins could hold the key to preserving long-term health and avoiding illness.
As humans continue their search for the fountain of youth, stem cells keep emerging as a key player in the quest for longevity. Studies increasingly suggest that preserving the fitness of stem cells leads to a longer healthspan, and new research highlights the importance of keeping stem cells clean and tidy.
According to a study recently published in the journal Cell Stem Cell, scientists at the University of California San Diego School of Medicine discovered that blood stem cells employ a surprising technique to eliminate their misfolded proteins. The researchers found that this process decreases with age and believe that boosting this specialized garbage disposal system could help fend off age-related illnesses.
The study focused on hematopoietic stem cells (HSCs), the cells in our bone marrow that produce new blood and immune cells throughout our lives. When their function is weakened or lost, this can lead to blood and immune disorders, such as anemia, blood clotting, and cancer.
UC San Diego researcher Robert Signer, Ph.D., describes how stem cells contribute to aging and age-related diseases. Credit: UC San Diego Health Sciences
“Stem cells are in it for the long haul,” said senior study author Robert Signer, Ph.D., associate professor at UC San Diego School of Medicine. “Their need for longevity requires them to be wired differently than all the short-lived cells in the body.”
A key to keeping stem cells happy is maintaining protein homeostasis. Previous work showed that stem cells, including HSCs, synthesize proteins much slower than other cell types, prioritizing quality over quantity. This helps them make fewer mistakes in the process, as misfolded proteins can become toxic to cells if allowed to build up.
Still, some mistakes or protein damage are inevitable, so the researchers set out to understand how stem cells ensure these proteins are properly discarded.
In most cells, damaged or misfolded proteins get individually tagged for disposal. A mobile protein destroyer called the proteasome then finds the labeled proteins and breaks them down into their original amino acid components. But in the new study, the researchers found proteasome activity was especially low in HSCs. This left the team puzzled: if getting rid of damaged proteins is so important to stem cells, why is the proteasome less active?
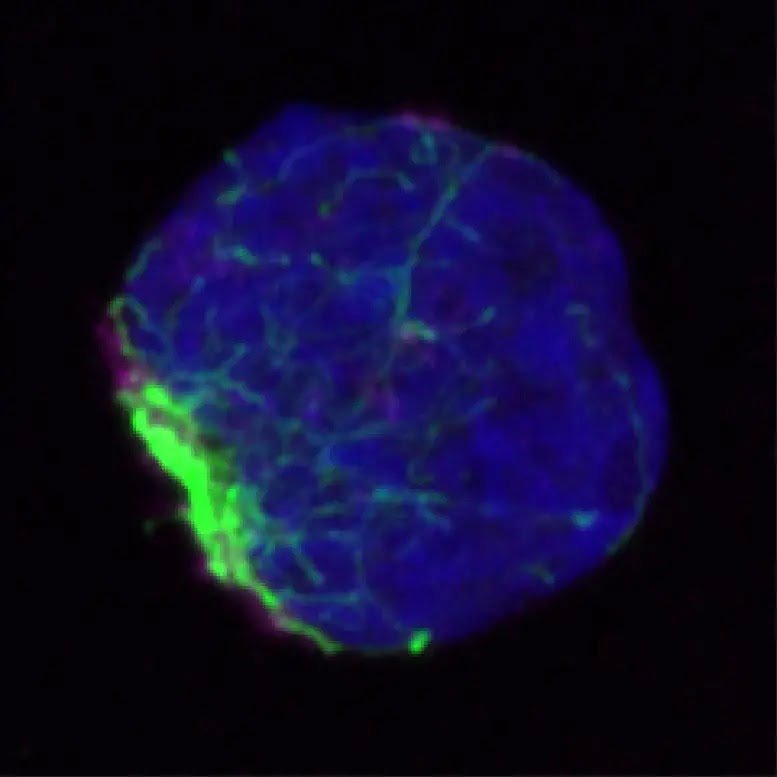
UC San Diego scientists found misfolded proteins were aggregated and caged into a single area (green) within stem cells before being disposed of. Credit: UC San Diego Health Sciences Through a series of subsequent experiments, the team discovered that HSCs use a different system entirely. Here, damaged and misfolded proteins are collected and trafficked into clusters called aggresomes. Once corralled into a single location, they can be collectively destroyed by the lysosome (a cell organelle containing digestive enzymes) in a process called aggrephagy.
“What’s very unusual here is this pathway was thought to only be triggered as an extreme stress response, but it’s actually the normal physiological pathway that’s used by stem cells,” said Signer. “This emphasizes how critical it is for stem cells to prevent stress so they can preserve their health and longevity.”
So why this different system? A main advantage of the proteasome method is that it breaks proteins down immediately, producing amino acids that the cell can reuse to build new proteins. But stem cells are less interested in building new proteins. Thus the authors suggest that by storing a collection of damaged proteins in one place, stem cells may be creating their own cache of resources that can be used at a later time when they are actually needed, such as after an injury or when it is time to regenerate.
“The body really can’t risk losing its stem cells, so having this stockpile of raw materials makes them more protected against rainy days,” said Signer. “Stem cells are marathon runners, but they also need to be world-class sprinters when the circumstances call for it.”
When the researchers genetically disabled the aggrephagy pathway, the stem cells started to accumulate aggregated protein, which impaired their fitness, longevity and regenerative activity.
The team then discovered that while almost all young stem cells had aggresomes, at a certain point in aging, they were almost completely gone. The authors suggest that stem cells’ inability to efficiently destroy misfolded proteins during aging is likely a key contributing factor to their declining function and the resulting age-related disorders.
“Our hope is that if we can improve stem cells’ ability to maintain the aggrephagy pathway, we will preserve better stem cell fitness during aging and mitigate blood and immune disorders,” said Signer.
The authors suspect that other types of stem cells and long-lived cells like neurons have a similar requirement for strict regulation of protein homeostasis, suggesting therapeutics to boost this pathway may be beneficial across multiple organs and pathologies.
Enter your journal: Reference: “Hematopoietic stem cells preferentially traffic misfolded proteins to aggresomes and depend on aggrephagy to maintain protein homeostasis” by Bernadette A. Chua, Connor J. Lennan, Mary Jean Sunshine, Daniela Dreifke, Ashu Chawla, Eric J. Bennett and Robert A.J. Signer, 21 March 2023, Cell Stem Cell.
DOI: 10.1016/j.stem.2023.02.010
Co-authors of this study include: Bernadette A. Chua, Connor J. Lennan, Mary Jean Sunshine, Daniela Dreifke and Eric J. Bennett at UC San Diego and Ashu Chawla at La Jolla Institute for Immunology.
Original Publisher : https://scitechdaily.com/clearing-out-the-trash-stem-cells-battle-against-aging/



